
Imaging modalities
RF-induced thermoacoustic tomography
Laser-induced photoacoustic tomography
Optical-coherence tomography (OCT)
Mueller optical-coherence tomography
Diffuse optical tomography (DOT)
Molecular and cellular bioimaging
Fluorescence microscopy
Scanning probe microscopy (SPM)
Scanning tunneling microscopy: its resolution is angstrom (atomic) level
Atomic force microscopy: its resolution is nanometer (macromolecular) level
Electron microscopy
Cryo-electron microscopy
Hyperpolarized xenon chemical exchange saturation transfer (HYPER-CEST)
Applications:
Knowledge base:
We learn from past history.
 |
NOBELFÖRSAMLINGEN KAROLINSKA INSTITUTET
THE NOBEL ASSEMBLY AT THE KAROLINSKA INSTITUTE
11 October 1979
The Nobel Assembly of Karolinska Institutet has decided today to award the
Nobel Prize in Physiology or Medicine for 1979 jointly to
Allan M Cormack and Godfrey Newbold Hounsfield
for the "development of computer assisted tomography".
An X-ray examination usually implies the passage of X-rays through an organ with
a resulting image of the organ on X-ray film. The dark areas on the film vary
according to the anatomy and the structure of the tissues being X-rayed.
A peculiarity of this picture is that it is two-dimensional. In the reproduction
the dimension of depth is lost. This means that an overall picture of the lungs,
for example, is a composite one in which all the details in the path of the rays
are overlapped. In order to acquire any depth perception, one must complement
frontal exposures with lateral exposures. The radiologist's interpretation of
possible changes in the lungs is based on his knowledge of the normal anatomy of
the lungs and of the properties of the pathological abnormalities. But the
nature of the final X-ray image makes judgement in certain cases undeniably
subjective. Therefore, in many situations there is a need to be able to isolate
the image of a section of an organ from the overlying structures by so-called
tomography (from the Greek tomos, a cut, and graph, written). Many
technical solutions have been tested during the course of the years but none
have been found to be entirely satisfying. For purely physical reasons one can
never achieve a complete eradication of other sections of the organ, and the
picture's contrast is reduced. This is true even when one allows the radiation
beam to run parallel to the examined section so that the rays proceed from one
edge to another. There are other limitations to conventional radiological
diagnostics. One is that X-rays cannot be utilized to more than 25 %;
another the X-ray film has a relatively low sensitivity in the reproduction of
the variations in tissue density.
In computer-assisted tomography these problems have been ingeniously solved.
When the method was introduced into medical care six years ago it quickly became
apparent that it signified something revolutionarily new, with great
repercussions with X-ray diagnostics and the medical disciplines that make use
of it.
The basic feature of the method is that the X-ray tube, in a definite pattern of
movement, permits the rays to sweep in many directions through a cross-section
of the body or the organ being examined. The X-ray film is replaced by sensitive
crystal detectors, and the signals emitted by amplifiers when the detectors are
struck by rays are stored and analyzed mathematically in a computer. The
computer is programmed to rapidly reconstruct an image of the examined
cross-section by solving a large number of equations including a corresponding
number of unknowns. The image presented on the screen of the oscilloscope is
drawn in a fine system of squares, a so-called matrix, in which each individual
square corresponds to a part of the examined organ. Each element expresses the
permeability of X-rays of the corresponding part of the organ. A fundamental
peculiarity is that the image elements do not influence each other while the
image is being reconstructed. In other words, there is no overlapping of
elements in the image. Because the sensitivity of the crystal detectors and
amplifier is more than 100 times as great as X-ray film, computed tomography can
detect very subtle variations of tissue density. This means that the density
resolution is exceptionally high. For all practical purposes one achieves a
correct image of a thin section of organ tissue.
The first computer tomograph was constructed to be used for examining the skull,
with special emphasis on diseases of the brain. The method soon experienced an
enormous breakthrough in the radiological diagnosis of neural diseases. The
reason is the precision and sensitivity of computed tomography. Extensive
special examinations, such as contrast encephalography and pneumaencephalography,
that is, X-ray examination with a contrast medium in the vessels, and filling
the brain cavities with air following lumbar puncture, provide very valuable
information, but nevertheless indirectly. The need for these types of
encephalography is now reduced. Computed tomography, on the other hand, provides
in each section a very detailed picture of the brain and its cavities as well as
the fluid-filled spaces surrounding the brain, i.e., the cisterns and
subarachnoid spaces, everything visible directly on the picture. This means that
pathological changes in the brain and its surroundings can be well demonstrated
by the computed tomogram. Their position, size and shape can be estimated and
their nature can often be determined. The number of black gradations in the
squared pattern of the image is greater than an observer can perceive, but can
be extracted from the image and denoted numerically. This appreciably simplifies
determination of the nature of the disease. Hemorrhages, and such changes in the
brain arising from a blood clot blocking circulation, cause the same symptoms
but can be distinguished by computed tomography. Tumors and conditions brought
on by inflammation, senile changes in the brain, hydrocephalus and malformations
in the brain can all be revealed. The method is invaluable in developing new
methods for operating on brain tumors. So rich is the detail that the computed
tomogram is reminiscent of the picture one gets of the brain at autopsy.
Computer-assisted tomography cause no discomfort to the patient, who lies
comfortably on his back during the examination. This makes it possible to
examine even very sick individuals in an acute phase of their illness. The
effect of the treatment can be monitored. All centers in the world with access
to a computed tomograph attest to the fact that the method has meant an enormous
advance in diagnostics, therapy, development and research within the specialty
of neurological diseases.
With modern computed tomographs it is possible to examine every organ in the
body. In certain connections the method is superior to all other methods. In
other situations it complements other techniques, such as ultrasound, isotope
diagnostics with the gamma camera.
A very important area of application, which is rapidly growing in importance, is
the radioactive treatment of tumors. Heretofore the weakest link in planning
radiation treatment has been the difficulty in determining, with desired
precision, the position, size and shape of tumors in the innermost regions of
the body. This involves the problem of delimiting tumors from surrounding
tissue. With computed tomography it is possible to carefully analyze all these
factors, from a scaled image of the body on a level with the tumor. This
facilitates the choice of suitable radiation field and optimal ray quality. When
the tumor shrinks during treatment, which can be shown by computed tomography,
the radiation can gradually be changed so that more resistant sections of the
tumor can be irradiated more intensely than surrounding tissue. Well-informed
observers believe that computer-assisted tomography has introduced a new era in
radiation therapy. The entire field is the subject of intensive research.
This year's Nobel Prize in physiology or medicine has been awarded to Allan M
Cormack and Godfrey N Hounsfield for their contributions toward the
development of computer-assisted tomography, a revolutionary radiological
method, particularly for the investigation of diseases of the nervous system.
Allan Cormack is professor and head of the institution of physics at
Tufts University in Medford,
Massachusetts, USA. He was the first, from a theoretical point of view, to
analyze the conditions for demonstrating a correct radiographic cross-section in
a biological system. He published his analysis of the problem in two scientific
publications in 1963 and 1964. He understood that the problem was basically a
mathematical one. It was a matter of finding a reasonable two-dimensional
function that described how X-rays attenuate in each individual part within a
slice when one knows the mean values of the rays' absorption, the so-called line
integrals, along a number of straight lines within this slice. He was convinced
that the problem had great principle interest and foresaw that, if it could be
solved, there would be possible applications within medicine, such as
radiotherapy and positron-camera diagnostics. He was not aware then that the key
mathematical problems had been considered earlier in an altogether different
connection and deduced his own method of calculation. In extensive model
experiments, in which he used gamma radiation that has a shorter wave-length
than X-rays, he showed that the agreement between theory and experiment was
good. Cormack's reconstruction mathematics is one of several possible ones that
can be used. His contributions to the development of the theory of
computer-assisted tomography was early and anticipated the coming development by
several years by being the first to state the principles for reconstructing a
cross-section of tissues in an organ based on these X-ray projections. The
reason Cormack's discovery did not come to be industrially applied is not known,
but it can be assumed that the computers of the time lacked sufficient capacity
to enable the method to be applicable to medical care.
Godfrey Hounsfield, who is chief of the medical research division of
Electric and Musical Industries, Middlesex, England, is the central figure in
computer-assisted tomography. He has made the really decisive contributions for
introducing computed tomography in medicine by constructing the first computed
tomography system practicable in medical care. Thus, he described a complete
system for computed tomography in his patent application in 1968. The patent was
granted in 1972. An advance communication about the method came in 1971, a more
extensive report with a supplement of clinical viewpoints by Ambrose followed in
1972, and a detailed description of the system appeared in the December, 1973,
issue of the British Journal of Radiology. This work and the patent papers are
epoch-making in medical radiology. The achievement is no less significant
because all the components forming the basis for the construction and operation
of the computed tomograph had been described earlier in non-medical
publications. Hounsfield was obviously unaware of Cormack's contributions and
developed his own method for reconstruction of the image. With an unusual
combination of vision, intuition and imagination, and with an extraordinarily
sure eye for the optimal choice of physical factors in a system that must have
offered very great problems to construct, he obtained results which in one blow
surprised the medical world. It can be no exaggeration to maintain that no other
method within X-ray diagnostics has, during such a short period of time, led to
such remarkable advances, with regard to research and number of applications, as
computer-assisted tomography.
Hounsfield's system, which was directed at examinations of the skull and brain,
started a development which in a few years led to the so-called fourth
generation of computed tomographs. In these, technical improvements and more
rapid analytical reconstruction methods have raised performance still farther,
work in which Hounsfield has taken active part.
 |
|
6 October 2003
The Nobel Assembly at Karolinska Institutet has today decided to award The
Nobel Prize in Physiology or Medicine for 2003
jointly to
Paul C Lauterbur and Peter Mansfield
for their discoveries concerning
"magnetic resonance imaging"
¡¡
Imaging of human internal organs with exact and non-invasive methods is very important for medical diagnosis, treatment and follow-up. This year's Nobel Laureates in Physiology or Medicine have made seminal discoveries concerning the use of magnetic resonance to visualize different structures. These discoveries have led to the development of modern magnetic resonance imaging, MRI, which represents a breakthrough in medical diagnostics and research.
Atomic nuclei in a strong magnetic field rotate with a frequency that is dependent on the strength of the magnetic field. Their energy can be increased if they absorb radio waves with the same frequency (resonance). When the atomic nuclei return to their previous energy level, radio waves are emitted. These discoveries were awarded the Nobel Prize in Physics in 1952. During the following decades, magnetic resonance was used mainly for studies of the chemical structure of substances. In the beginning of the 1970s, this year¡¯s Nobel Laureates made pioneering contributions, which later led to the applications of magnetic resonance in medical imaging.
Paul Lauterbur (born 1929), Urbana, Illinois, USA, discovered the possibility to create a two-dimensional picture by introducing gradients in the magnetic field. By analysis of the characteristics of the emitted radio waves, he could determine their origin. This made it possible to build up two-dimensional pictures of structures that could not be visualized with other methods.
Peter Mansfield (born 1933), Nottingham, England, further developed the utilization of gradients in the magnetic field. He showed how the signals could be mathematically analysed, which made it possible to develop a useful imaging technique. Mansfield also showed how extremely fast imaging could be achievable. This became technically possible within medicine a decade later.
 |
| MRI is used for imaging of all organs in the body. |
Magnetic resonance imaging, MRI, is now a routine method within medical diagnostics. Worldwide, more than 60 million investigations with MRI are performed each year, and the method is still in rapid development. MRI is often superior to other imaging techniques and has significantly improved diagnostics in many diseases. MRI has replaced several invasive modes of examination and thereby reduced the risk and discomfort for many patients.
Water constitutes about two thirds of the human body weight, and this high water content explains why magnetic resonance imaging has become widely applicable to medicine. There are differences in water content among tissues and organs. In many diseases the pathological process results in changes of the water content, and this is reflected in the MR image.
Water is a molecule composed of hydrogen and oxygen atoms. The nuclei of the hydrogen atoms are able to act as microscopic compass needles. When the body is exposed to a strong magnetic field, the nuclei of the hydrogen atoms are directed into order ¨C stand "at attention". When submitted to pulses of radio waves, the energy content of the nuclei changes. After the pulse, a resonance wave is emitted when the nuclei return to their previous state.
The small differences in the oscillations of the nuclei are detected. By advanced computer processing, it is possible to build up a three-dimensional image that reflects the chemical structure of the tissue, including differences in the water content and in movements of the water molecules. This results in a very detailed image of tissues and organs in the investigated area of the body. In this manner, pathological changes can be documented.
The resonance phenomenon is governed by a simple relation between the strength of the magnetic field and the frequency of the radio waves. For every type of atomic nucleus with unpaired protons and/or neutrons, there is a mathematical constant by which it is possible to determine the wavelength as a function of the strength of the magnetic field. This phenomenon was demonstrated in 1946 for protons (the smallest of all atomic nuclei) by Felix Bloch and Edward Mills Purcell, USA. They were awarded the Nobel Prize in Physics in 1952.
Other fundamental discoveries concerning magnetic resonance have in recent years resulted in two Nobel Prizes in Chemistry. In 1991, Richard Ernst, Switzerland, was awarded for his contributions to the development of the methodology of high resolution nuclear magnetic resonance spectroscopy. In 2002, Kurt W¨¹thrich, also Switzerland, was awarded for his development of nuclear magnetic resonance spectroscopy for determination of the three-dimensional structure of biological macromolecules in solution.
This year's Nobel Laureates in Physiology or Medicine are awarded for crucial achievements in the development of applications of medical importance. In the beginning of the 1970s, they made seminal discoveries concerning the development of the technique to visualize different structures. These findings provided the basis for the development of magnetic resonance into a useful imaging method.
Paul Lauterbur discovered that introduction of gradients in the magnetic field made it possible to create two-dimensional images of structures that could not be visualized by other techniques. In 1973, he described how addition of gradient magnets to the main magnet made it possible to visualize a cross section of tubes with ordinary water surrounded by heavy water. No other imaging method can differentiate between ordinary and heavy water.
Peter Mansfield utilized gradients in the magnetic field in order to more precisely show differences in the resonance. He showed how the detected signals rapidly and effectively could be analysed and transformed to an image. This was an essential step in order to obtain a practical method. Mansfield also showed how extremely rapid imaging could be achieved by very fast gradient variations (so called echo-planar scanning). This technique became useful in clinical practice a decade later.
The medical use of magnetic resonance imaging has developed rapidly. The first MRI equipments in health were available at the beginning of the 1980s. In 2002, approximately 22 000 MRI cameras were in use worldwide, and more than 60 million MRI examinations were performed.
A great advantage with MRI is that it is harmless according to all present knowledge. The method does not use ionizing radiation, in contrast to ordinary X-ray (Nobel Prize in Physics in 1901) or computer tomography (Nobel Prize in Physiology or Medicine in 1979) examinations. However, patients with magnetic metal in the body or a pacemaker cannot be examined with MRI due to the strong magnetic field, and patients with claustrophobia may have difficulties undergoing MRI.
Today, MRI is used to examine almost all organs of the body. The technique is especially valuable for detailed imaging of the brain and the spinal cord. Nearly all brain disorders lead to alterations in water content, which is reflected in the MRI picture. A difference in water content of less than a percent is enough to detect a pathological change.
In multiple sclerosis, examination with MRI is superior for diagnosis and follow-up of the disease. The symptoms associated with multiple sclerosis are caused by local inflammation in the brain and the spinal cord. With MRI, it is possible to see where in the nervous system the inflammation is localized, how intense it is, and also how it is influenced by treatment.
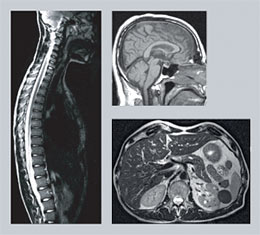 |
| Examination with MRI is especially valuable for detailed imaging of the brain and the spinal cord. |
Another example is prolonged lower back pain, leading to great suffering for the patient and to high costs for the society. It is important to be able to differentiate between muscle pain and pain caused by pressure on a nerve or the spinal cord. MRI examinations have been able to replace previous methods which were unpleasant for the patient. With MRI, it is possible to see if a disc herniation is pressing on a nerve and to determine if an operation is necessary.
Since MRI yields detailed three-dimensional images, it is possible to get distinct information on where a lesion is localized. Such information is valuable before surgery. For instance, in certain microsurgical brain operations, the surgeon can operate with guidance from the MRI results. The images are detailed enough to allow placement of electrodes in central brain nuclei in order to treat severe pain or to treat movement disorders in Parkinson's disease.
MRI examinations are very important in diagnosis, treatment and follow-up of cancer. The images can exactly reveal the limits of a tumour, which contributes to more precise surgery and radiation therapy. Before surgery, it is important to know whether the tumour has infiltrated the surrounding tissue. MRI can more exactly than other methods differentiate between tissues and thereby contribute to improved surgery.
MRI has also improved the possibilities to ascertain the stage of a tumour, and this is important for the choice of treatment. For example, MRI can determine how deep in the tissue a colon cancer has infiltrated and whether regional lymph nodes have been affected.
MRI can replace previously used invasive examinations and thereby reduce the suffering for many patients. One example is investigation of the pancreatic and bile ducts with contrast media injection via an endoscope. This can in some cases lead to serious complications. Today, corresponding information can be obtained by MRI.
Diagnostic arthroscopy (examination with an optic instrument inserted into the joint) can be replaced by MRI. In the knee, it is possible to perform detailed MRI studies of the joint cartilage and the cruciate ligaments. Since no invasive instrument is needed in MRI, the risk of infection is eliminated.
Reflections on MRI - leadership reflections
This paper appears in:
Signal Processing Magazine, IEEE
Publication Date: Jan. 2005
Volume: 22, Issue: 1
On page(s): 12- 13
¡¡
A dramatic example of terahertz imaging is provided by ESA.
By observing microwave radiation it is possible to map the various properties of
the Earth's environment. The image below shows the sea surface temperature as
measured by the ATSR instrument on the
![]() ERS
satellites.
ERS
satellites.
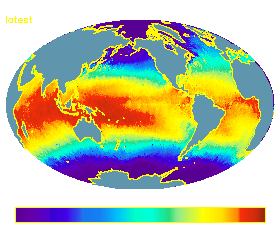
ATSR - image of sea surface temperature, February 2002
Another dramatic example of terahertz imaging has been
provided by
![]() Qinetiq
who are pioneering the development of compact millimetre wave imaging systems.
The image below was produced with one of their systems and shows the interesting
properties of terahertz waves.
Qinetiq
who are pioneering the development of compact millimetre wave imaging systems.
The image below was produced with one of their systems and shows the interesting
properties of terahertz waves.

Image taken with a mm wave camera
click for animation (AVI, 293Kb), courtesy of Qinetiq
Terahertz imaging is fast being recognised as a powerful diagnostic technique.
Terahertz imaging can be achieved by observing the natural terahertz waves emitted by pretty much everything. Unlike light, terahertz waves are able to propagate through cloud and smoke providing a powerful advantage for certain remote sensing measurements. From a practical aspect they are also able to pass through windows, paper, clothing and in certain instances even walls.
There are many possible imaging applications for the
technology
![]() will
provide. If successful, such an imaging array will represent a true breakthrough
for terahertz remote sensing from Space (one of ESA's primary interests) and
will have dramatic implications for the next generation of Astronomy and Remote
sensing systems.
will
provide. If successful, such an imaging array will represent a true breakthrough
for terahertz remote sensing from Space (one of ESA's primary interests) and
will have dramatic implications for the next generation of Astronomy and Remote
sensing systems.
The
![]() imager will operate at frequencies of 250 and
300 GHz which should dramatically reduce the volume and mass of the imager. The
use of two frequencies provide a means for contrasting between materials with
different transmission and reflection properties, effectively creating two
colours. For remote sensing the detection of more than one frequency would allow
the simultaneous measurement of two atmospheric constituents
imager will operate at frequencies of 250 and
300 GHz which should dramatically reduce the volume and mass of the imager. The
use of two frequencies provide a means for contrasting between materials with
different transmission and reflection properties, effectively creating two
colours. For remote sensing the detection of more than one frequency would allow
the simultaneous measurement of two atmospheric constituents
There are also likely to be commercial and technology off-shoots in the following sectors:
As terahertz waves are also able to penetrate the uppermost layers of skin making the early detection of skin cancers an exciting possibility. The unique properties of terahertz waves mean that there will undoubtedly be many other new applications; how many we just don't know as the technology simply isn't there... YET!
¡¡